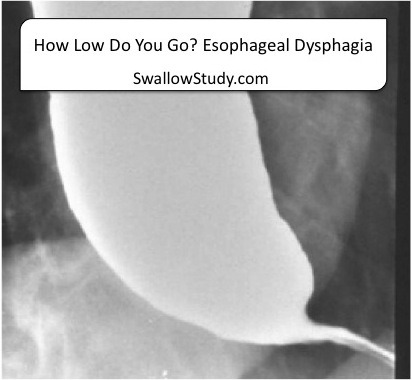
You do not diagnose what this is, but the SLP comments on bolus clearance. This barium is not clearing and could cause retrograde flow and aspiration.
by Karen Sheffler, MS, CCC-SLP, BCS-S of SwallowStudy.com
This blog was inspired by: “Esophageal Screening: The Essential Last Step in Your Dysphagia Evaluation,” by Ashli O’Rourke, MD, April 13, 2014, at ASHA’s Health Care & Business Institute in Las Vegas, NV. It was particularly special to hear her perspective, because she started her career as an SLP and is now an Otolaryngologist specializing in adults with problems of the larynx and esophagus. She works at the Evelyn Trammell Institute for Voice and Swallowing at MUSC.
“We tend to stop at the cervical esophagus,” lamented Dr Ashli O’Rourke. “Esophageal dysphagia is this orphan out there.”
Let’s imagine a case: a man comes in for an outpatient Videofluoroscopic SwallowStudy.com (VFSS), aka, Modified Barium SwallowStudy.com (MBSS) with complaints that pills, breads and meats are getting stuck. He vaguely points to the suprasternal through retrosternal region. He is assessed in a lateral position to the level of the cervical esophagus with liquids, barium pudding and a cookie. His oropharyngeal phases are normal. No residue was seen in the pharynx to explain his problem. The report concludes that it must be a globus sensation. Have we really answered all his concerns? Have we even tested items that typically get stuck? If we did do an esophageal screen on our MBSS with liquids only, are we fully evaluating his problem?
Reasons to include the esophagus in your evaluation of swallowing:
- It is in our Scope of Practice. Check out ASHA’s Scope of Practice in Speech-Language Pathology (2007): https://www.asha.org/policy/SP2007-00283/#sec1.6
- The bolus does not stop at the cervical esophagus.
- Interrelationships between the pharyngeal and esophageal phases: a problem in one area will affect the other. Gulling, et al. (2012) found 82% of patients with a delay in initiation of pharyngeal swallow per MBSImP also had impaired clearance through the esophagus. If your swallow is weak and slow to start, then it may be weak and slow throughout.
- It is better for our patients to provide a comprehensive evaluation. Our thorough history taking and our bedside swallow exam often can point to the esophagus as the potential source of the dysphagia.
- Our comprehensive evaluations do not diagnose issues in the esophagus, but they can aide in differential diagnosis by noting suspected esophageal motility versus obstructive esophageal issues. We can make timely and appropriate referrals.
- Aspiration after the swallow may be due to stasis or retention in the esophagus with supraesophageal reflux (also known as a retrograde flow or esophageal backflow) into the laryngeal vestibule. This can be refluxed material getting up high, also known as supraesophageal reflux disease (SERD) or laryngopharyngeal reflux (LPR), which are different from gastroesophageal reflux. (Please see this article on types of reflux and treatments for more information.)
- Treatments for a pharyngeal problem can affect the esophagus. The effortful swallow may help distal esophageal clearance. However, the strategies of double swallow to clear residue in the throat, valsalva, and the Mendelsohn can negatively affect the bolus flow through the esophagus. For example: the double swallow, if done within 10 seconds of the primary swallow, can halt the secondary peristaltic wave in the esophagus (this is a necessary cleaning wave triggered neurologically by the distention of the esophagus itself). Per O’Rourke, evidence from HRM (High Resolution Manometry) shows to wait at least 10 seconds after the first swallow before performing a dry swallow. We are balancing risks of pharyngeal residue versus risks of esophageal dysphagia, but the main issue is to make informed treatment recommendations.
“80% of esophageal disorders can be diagnosed by history alone,” per O’Rourke.
Again, WE are NOT the ones who diagnose, but with the time we spend on obtaining a thorough history, we can find clues to esophageal dysphagia.
8 Potential Clues to Esophageal Dysphagia on your Clinical Exam:
1. Retrosternal localization of food/liquid getting stuck (or more distal localization).
- Clarify if it is a dysphagia to solids/liquid/pills, or all three.
- Have the patient point exactly where.
- Hint: motility problems in the esophagus tend to cause dysphagia to liquids and solids, whereas as an obstructive/mucosal issue (i.e., stricture) tends to cause dysphagia to solids (i.e., breads, meats, and large pills).
- Note: Roeder, et al (2004) found that the patient’s distal localization tends to be accurate about 80% of the time. I have seen a patient point right to the food above a stricture on my esophageal screening during an MBSS. However, Roeder’s study showed that out of the 55 patients who pointed to the proximal esophagus (retrosternal), only 1 of them actually had a problem at the level of the proximal esophagus. I have frequently heard doctors wonder if there is a Zenker’s Diverticulum, but these are fairly rare. More likely, if the patient is pointing to the cervical/proximal esophageal area, the problem may be somewhere in the esophagus, but probably distal to that exact location. Hence, we need to scan the esophagus fully on our MBSS.
2. The stuck food eventually passes and/or passes with liquids.
- Does a liquid wash work? If it does, then maybe it is a stricture or another esophageal obstructive issue.
- Does the liquid also come back up? Liquid regurgitation may point to an esophageal dysmotility. OR if a stricture is bad enough to cause a food impaction in the esophagus, then even liquid and saliva cannot make it through. If you see someone expectorating buckets of saliva, and it is not a neurological etiology, then think food impaction.
3. Odynophagia
- Have the patient rate the pain on a scale of 1-10 and localize the pain.
- It could be as simple as thrush (esophageal candidiasis) or bad esophagitis from reflux.
- Pain could also be a partial denture plate lodged in the bottom of the throat. Yes, please ask when the dentures were last seen! I have seen several people who had swallowed partial plates overnight while sleeping. Those metal prongs really can get stuck in the throat.
- However, pain with swallowing, plus dysphagia to solids, plus weight loss needs a rapid referral to GI with suspicion of esophageal cancer.
- Is there non-cardiac chest pain and does it radiate to the back? Refer to GI for suspicion of an esophageal dysmotility disorder (e.g., achalasia).
4. Globus sensation. Have the patient point where.
5. Regurgitation, reflux, “vomiting”
- Specify true nausea/vomiting versus a return of the liquid/solid bolus back up to the throat after the swallow.
- Differentiate descriptions of backflow of food/liquid out of the esophagus versus gastroesophageal reflux and supraesophageal reflux versus vomiting due to another GI issue (i.e., bowel obstruction).
- Ask about frequency and timing of regurgitation and coughing
- Clarify sputum characteristics of stringy, frothy, containing food, tinged with a liquid (GERD/SERD patients tend to have frothy sputum in the morning)
- Is there undigested food/liquid in the regurgitation?
6. Halitosis. Long-term stasis of food in the esophagus may cause bad breath.
7. Coughing well after the swallow or post-prandial coughing (after eating a meal)
8. Early satiety (feeling of getting full quickly)
- If the patient eats only a small bowl of soup and is full, then a potential source is delayed or absent esophageal clearance. The MD would also look into a gastric outlet obstruction and delayed gastric emptying. For example, delayed gastric emptying can occur in patients with Diabetes.
Common SLP instrumental evaluations that can suggest esophageal dysphagia:
1. Clues from a FEES/FEESST exam:
- Post-cricoid residue with the bolus or frothy secretions.
- Pooling of the bolus and secretions in the pyriforms.
- Regurgitation of the bolus out of the esophagus after the swallow.
- Endoscopic signs of mucosal irritation, such as edema and erythema to the arytenoids and interarytenoid space. This may suggest laryngopharyngeal reflux, but the SLP does not diagnose this. We can make appropriate referrals to an ENT and/or GI physician.
2. Clues from a MBSS with Esophageal Follow-Through:
- Main Goal: Observe esophageal clearance with the aide of gravity.
- Think about how the physiology affects the bolus flow. Did the bolus pass through or not?
- This is a screening, where the purpose is not to specifically measure the timing or make a diagnosis. Your report can refer to the radiologist’s report for further descriptions.
- Observe the opening, distention and duration of opening of the cricopharyngeus muscle (at the pharyngoesophageal segment/upper esophageal sphincter). Does it obstruct the bolus flow?
- Continue with the esophageal follow-through to the stomach to observe the esophageal emptying through the LES.
- If a bolus remains in the esophagus (stasis), the SLP can note if there is aspiration or a risk for aspiration after the swallow due to a retrograde flow.
- Use a viscous bolus – test purees with at least 2 trials, per O’Rourke. If you give a “lazy bolus” like thin liquid, you will not fully challenge the esophagus. A viscous bolus requires increased contractile forces with increased pressures for clearance. A high density barium is also useful in the esophagus (which is a collapsed system) to see mucosal abnormalities, per O’Rourke. Low density, thin liquid barium, can be used to pick up rings and webs if the esophagus can be well-distended with large volumes. I would add, if the patient is having trouble with pills, then test pills. If the patient is having trouble with bread, then test bread. Bread acts very differently in the esophagus than a smooth barium pudding.
Jou, et al (2009, section #5) stated: “In our normal cohort, semisolid boluses elicited significantly more stasis than liquid boluses. This suggests that a variety of barium product consistencies simulating food-like viscosities need to be used, in addition to traditional fluids, to fully evaluate esophageal transport with the videofluoroscopic esophagram. Our protocol included observations of esophageal motility with semisolid barium which has not been documented previously. Modification of the esophagram protocol may increase the sensitivity of detecting abnormal esophageal motility in those patients who are symptomatic.”
- Are there functional strategies to improve the clearance (i.e., avoid the rapid double swallow, chew well, liquid wash, lubricate the system if xerostomia, effortful swallow, slow rate, small-frequent meals, diet modifications, etc.)? I’ve used the trick of ginger-ale to get a stuck pill to clear through the esophagus. Keep in mind that the barium tablets have a hydrostatic quality and can stick to mucosa in a normal patient. Instead, the exam with a large barium pill is looking for an obstruction hindering the bolus flow?
- Note: stasis at the aortic arch is normal, per O’Rourke. This is the transition zone with a low pressure gap of about 2 centimeters in the supine position per research from HRM, where the striated proximal 1/3rd of the esophagus transitions to the smooth muscle distal 2/3rds. In a seated position, the gap may be slightly larger and still allow for complete bolus clearance (Park, et al., 2014).
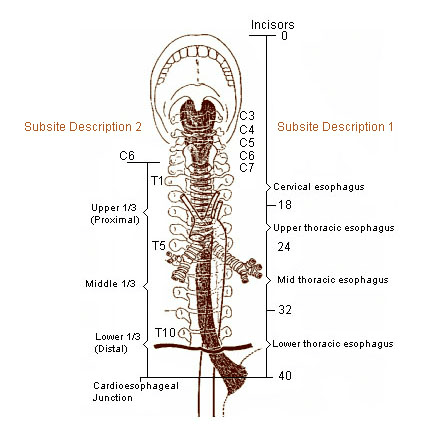
See labels indicating location of proximal 1/3 to distal 1/3 of esophagus.
- I recall Dr Joanne Robbins discussed the need for a videoflouroscopic esophagram in our dysphagia course back in the early 1990’s at University of Wisconsin-Madison. More recently, Robbins has used the term oroesophagram in lectures. Here is a link to the Jou, et al (2009) pilot study she was involved with: Esophageal Clearance Patterns in Normal Older Adults as Documented with Videofluoroscopic Esophagram https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2752658/
- Watch that the patient does not double swallow immediately as you are following the bolus through the esophagus.
- Research using HRM (High Resolution Manometry) has found that it is within normal limits to have a failure of contraction in the esophagus for up to 3 out of 10 swallows. This is why scanning down the esophagus on several boluses is necessary. You can note if the stasis and delay is intermittent or consistent.
- Gullung, et al (2012) noted an abnormal Esophageal Clearance* on the MBSImP correlated strongly with abnormal esophageal findings on HRM (High Resolution Manometry – described as “multichannel intraluminal impedance-esophageal manometry” in this study).
*Esophageal Clearance is component #17 on the MBSImP (Martin-Harris, et al., 2008). The clinician rates it as:
-
-
- complete,
- esophageal retention,
- esophageal retention with retrograde flow below the PES (pharyngoesophageal segment),
- esophageal retention with retrograde flow through the PES, or
- minimal to no esophageal clearance.
-
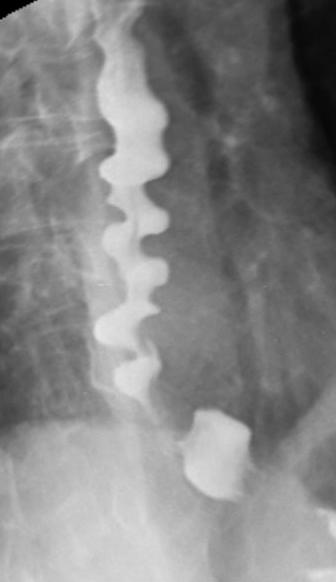
This would be an example of minimal to no clearance of the bolus.
O’Rourke recommended the WGO Practice Guidelines as a method to categorize esophageal issues: https://www.worldgastroenterology.org/assets/downloads/en/pdf/guidelines/08_dysphagia.pdf
Here is an excerpt from page 6 of the WGO Practice Guidelines for Dysphagia:
Most common causes of esophageal dysphagia:
1. Intraluminal foreign bodies (usually a cause of acute dysphagia)
2. Mucosal diseases (intrinsic structural/obstructive issues):
-
-
- Peptic stricture secondary to gastroesophageal reflux disease
- Esophageal rings and webs
- Esophageal tumors
- Chemical injury (e.g., caustic ingestion, pill esophagitis, sclerotherapy for varices)
- Radiation injury
- Infectious esophagitis
- Eosinophilic esophagitis
-
3. Mediastinal diseases (extrinsic)
-
-
- Tumors (e.g., lung cancer, lymphoma)
- Infections (e.g., tuberculosis, histoplasmosis)
- Cardiovascular (dilated auricula, vascular compression)
-
4. Diseases affecting smooth muscle and its innervation
-
-
- Achalasia (both idiopathic and associated with Chagas disease)
- Scleroderma
- Other motility disorders
- Post-surgical (i.e., after fundoplication, antireflux devices)
-
Conclusions on Esophageal Dysphagia:
I hope this blog is a useful resource on esophageal dysphagia. I have had a special place in my heart for the “orphaned” esophagus since beginning my practice as a Speech-Language Pathologist in 1995. This may be due to the influence of the midwest, studying at University of Wisconsin-Madison and discussing the esophagus with Dr Reza Shaker of Milwaukee. I have made observations with esophageal follow-throughs on Modified Barium Swallows Studies since 1999. Going forward, let there be no debate about whether or not the esophagus is in our scope of practice. It is a crucial part of the comprehensive swallowing evaluation. Let’s all make sure the esophagus is not the “orphaned” part of our patients’ bodies!
A brief and final word on HRM:
I would love to discuss HRM (High Resolution Manometry) further, but check out the 3rd issue discussed in this Dysphagia Research Society Annual Meeting review blog and the resource below. HRM presents the swallow pressures as a rainbow of colors to represent the pressures in the pharynx through the LES. With the addition of impedance testing (pressure exerted on the sensors by the bolus), we can “see” the bolus in purple! How did they know purple was my favorite color!
For a start, here is a link to the Park, et al (2014) article on High-Resolution Impedance Manometry that includes pictures and good references. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4204409/
*******
New Esophageal Dysphagia Resource on SwallowStudy.com:
Dysphagia Digest: Multiphase Dysphagia Evaluations & Pharyngoesophageal Inter-Relationships
References cited per O’Rourke’s talk:
Gullung, J.L., Hill, E.G., Castell, D.O. & Martin-Harris, B. (2012). Oropharyngeal and esophageal swallowing impairments: Their association and the predictive value of the modified barium swallow impairment profile and combined multichannel intraluminal impedance-esophageal manometry. Annals of Otology, Rhinology & Laryngology, 121 (11): 738-745.
Roeder, B.E., Murray, J.A. & Dierkhising, R.A. (2004). Patient localization of esophageal dysphagia. Digestive Diseases and Sciences, 49 (4): 697-701.
Other References:
Jou, J., Radowsky, J., Gangnon, R., Sadowski, E., Kays, S., Hind, J., Gaumnitz, E., Taylor, A. & Robbins, J. (2009). Esophageal clearance patterns in normal older adults as documented with videoflouroscopic esophagram. Gastroenterology Research and Practice, September, 23, 2009. doi: 10.1155/2009/965062
Martin-Harris, B., Brodsky, M.B., Michel, Y., Castell, D.O., Schleicher, M., Sandidge, J., Maxwell, R. & Blair, J. (2008). MBS measurement tool for swallow impairment – MBSImP: Establishing a standard. Dysphagia, 23: 392-405.
Park, E.J., Lee, J.S., Lee, T.H., Bok, G.H., Hong, S.J., Kim, H.G., Jeon, S.R. & Kim, J. (2014). High-resolution impedance manometry criteria in the sitting position indicative of incomplete bolus clearance. Journal of Neurogastroenterology and Motility, 20 (4): 491-496. doi: 10.5056/jnm14012


Thank you!